SODI-IVIDIL
Imagine squirting a drop of coloured dye—say, red—into a glass of water. What happens? One starts with a clear glass of water with a drop of red dye in it, sees a red swirl spread, and ends with a glass of reddish water.
This process is known as diffusion: the random, continuous motion of molecules in a liquid or gas mixture that causes certain trends in the molecules' movement. These trends include, among others, moving from a higher to a lower concentration, or from a hot to a cold area. (Without diffusion, heaters would have a much harder time doing their job in winter!).

Dr. Ziad Saghir of Ryerson University.
Diffusion due to temperature is called thermodiffusion. The "Soret effect" is what happens when a liquid mixture contains a temperature gradient (exactly like a colour gradient, only with different temperatures instead of colours at each end). Canadian scientist Ziad Saghir, from Ryerson University, has been studying thermodiffusion in liquid mixtures for fifteen years. He intends to further his investigation--as far, in fact, as the International Space Station (ISS).
Saghir is part of a group of European and Russian scientists who make up the SODI-IVIDIL team. It is coordinated by Prof. V. Shevtsova from Microgravity Research Center (MRC), Université Libre de Bruxelles (ULB). The Belgian team (MRC, ULB) is responsible for the development and implementation of the experiment, while the numerical and theoretical support is shared between all participants (MRC, Belgium; Ryerson University, Toronto, Canada and a Russian team from Perm). The original idea for this research came from an international research competition. Following this competition, the European Space Agency developed the equipment required to do the experiment on the ISS.
This international team of scientists has been lured to the ISS because of one very important feature: microgravity. On Earth, gravity gives molecules weight, and the different weights between different molecules cause "buoyancy-driven" convection currents that also cause molecular movement and disrupt diffusion. Furthermore, the more liquids in one mixture, the more instability it has. Thus, to properly learn about how thermal diffusion works, it is necessary to isolate it so that it is the only thing affecting molecular movement in a liquid—gravity must be removed. SODI-IVIDIL aims to do just that.

A scientist inspects the insert known as IVIDIL situated in the SODI facility and Microgravity Science Glovebox. (Credit: ESA)
The hardware developed by Verhaert Space NV (Kruibeke, Belgium), SODI ("Selectable Optical Diagnostic Instrument") examines the effect of vibrations on thermodiffusion. IVIDIL is a vibrating bench apparatus on which a liquid sample is placed. As the amplitude (strength) and frequency of vibrations are changed, SODI will collect data on the mixture's temperature, concentration gradient, and progression of diffusion with time.
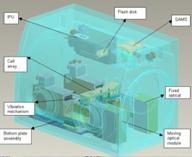
The SODI-IVIDIL components. (Credit: ESA)
Those who drive cars may appreciate this study: Saghir hopes to apply their findings to improving techniques in the oil industry. More specifically, oil wells contain mixtures of different hydrocarbon components having various molecular weights. Due to the Soret effect, oil sampling may change from one location to another in oil reservoirs: heavier substances rise, while lighter ones sink. Based on previous studies, the science team has developed numerical simulations to help understand the oil behaviours in a given well. The SODI-IVIDIL experiment will allow them to confirm and refine the parameters of their models, leading to more accurate predictions about oil wells being considered for extraction.
Canadian astronaut Dr. Robert (Bob) Thirsk, with Belgian crewmate Frank DeWinne, assembled both SODI and IVIDIL as well as conducted the experiment during their Expedition 20/21 mission, launched in May 2009. Dr. Thirsk also replaced IVIDIL with a second Canadian experiment using SODI, the "Diffusion Soret Coefficient" (DSC) study, which examined diffusion in six different liquids over time in microgravity.